Review / mini-review: Expert Opinion on Emerging Drugs
A guide for authors submitting to the Expert Collection
Scope
Expert Opinion on Emerging Drugs was devised to provide an expert evaluation of the most important emerging drugs in Phase II and III clinical trials. These are assessed in terms of their potential impact on the current management of specific diseases.
Many sources of information on compounds in development are available to research and development decision makers, but none of these provides an accessible and independent assessment of the commercial and technical viability of the most promising areas of drug research. Expert Opinion on Emerging Drugs seeks to fulfil this function by commissioning recognised opinion leaders to write focused appraisals of areas of innovative medical research, which can be readily understood by a scientifically-literate, but not necessarily expert, audience.
Each issue contains chapters on a variety of subjects, each of them profiled according to the standard format outlined below. The chapters are expected to be able to stand alone in terms of their coverage of each individual area, but their uniformity, in terms of the sections covered in each of the chapters, is designed to facilitate comparative evaluation.
The ideal Review Article will provide both a critical evaluation and the author’s opinion on the quality and novelty of the information disclosed.
Word limit
The word limit for Review Articles is 5,000-7,000 words (not including figures, tables or references).
Explore the Expert Collection
Discover all Expert Collection, journal specific guidelines.
Every article must contain
Please note that only the address of the first author of the article will appear on Medline/PubMed, not necessarily the corresponding author.
The aim of the abstract is to draw in the interested reader and provide an accurate reflection of the content of the paper. We therefore request the following structure is followed for full-length review articles:
Introduction: Authors are required to describe the significance of the topic under discussion.
Areas covered: Authors are required to describe the research discussed and the literature search undertaken.
Expert opinion: Authors are required to summarise briefly their Expert opinion section.
References must not be included in the abstract.
Medical need: This section should focus on the need for alternative/improved remedies for specific indications. For example, the limited palliative relief provided to arthritis patients by NSAIDs and the frequent impracticalities of surgery in a typically ageing population, highlight the need for genuine disease modifying therapy.
Existing treatment: This may be included as either a subsection of Medical need, or as a separate section, discussing the short-comings of currently-available therapy and where there are significant problems, e.g., side-effects, dosing regimens, lack of efficacy, etc.
Market review: A logical sequel to the above sections and an introduction to the next is a quantification of the current market size broken down in terms of territories, for the particular indication/drug category being discussed (see sample chapter provided). An estimation of the annual growth should also be included. These data should be tabulated, both for emphasis and brevity; comments should be limited to issues likely to affect the market acceptance of, or readiness for, particular therapeutic proposals. It is important to authenticate the source of the information quoted. If possible, a table of currently available compounds (or drug types), together with their market share should be included. The Editor may be able to obtain some data on a case-by-case basis.
Current research goals: This section should summarise briefly the aims of current research into the therapeutic area or drug class in question, as a prelude to the next section.
Scientific rationale: Please describe clearly any complex scientific concepts, so that readers are able to understand the basic principles of the specific disease target and thereby decide whether the proposal is scientifically tenable. The scientific hypothesis can form the basis for the prediction or anticipation of adverse events to which attention should be drawn. Including an illustration to aid understanding is recommended.
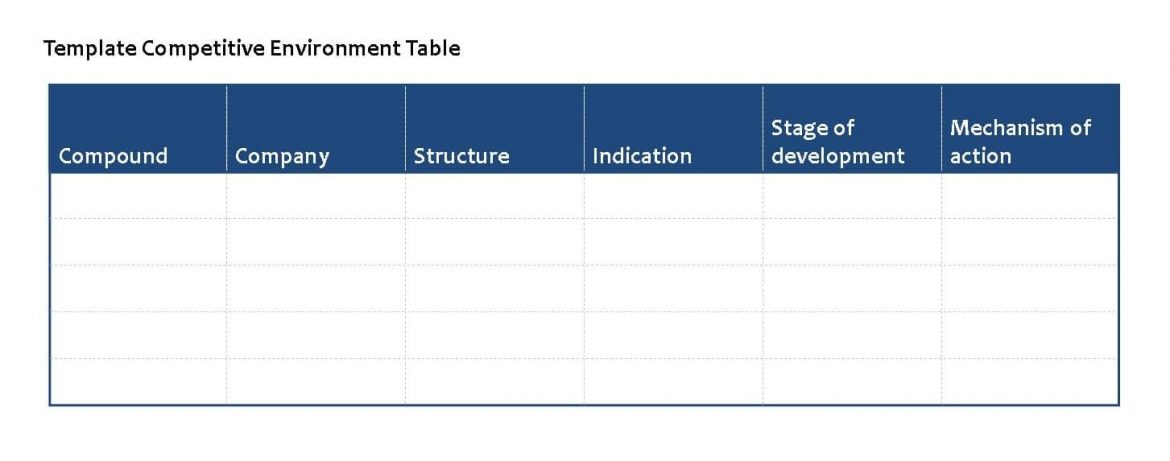
Competitive environment: This section should review specific compounds with claims for activity in the therapeutic target involved. The products selected would normally be derived from the literature or Pharmaprojects, although other substantiated sources may be accessed. The aim of this section is to demonstrate clearly and succinctly what compounds are in development for a given indication, by whom they are being developed and what stage they have reached in development. Authors should provide their comments in the form of evaluated and considered opinions on the prospects for this particular drug. Any additional data should be included here, e.g., potency, side-effects, likely success, etc.
Each review should include a table following the template provided below. Authors will be provided with information from Pharmaprojects to complete this section.
Potential development issues: These may arise as a consequence of the reviews above (e.g., class-specific, pharmacologically-linked adverse events) or be consequential of particular chemical structures. Alternatively, social issues and those of high public interest or comparatively elusive patient populations may influence the attractiveness of a given area. Existing orphan drug studies should be indicated where appropriate.
Conclusion: The conclusion for all articles should contain a brief summary of the data presented in the article. Please note that this section is meant to be distinct from, and to appear before the ‘Expert opinion’ section.
– What are the key findings and weaknesses in the research done in this field so far?
– What potential does this research hold? What is the ultimate goal in this field?
– What research or knowledge is needed to achieve this goal and what is the biggest challenge in this goal being achieved?
– Where do you see the field going in the coming years? What is going to happen?
– Which drugs discussed in this paper hold the most promise?
– Is there any particular area of the research you are finding of interest at present?
Please note that ‘opinions’ are encouraged in the Expert Opinion section, and, as such, referees are asked to keep this in mind when peer reviewing the manuscript.