Registered Reports at Taylor & Francis
Explore journals offering Registered Reports
What is a Registered Report?
The Center for Open Science describes Registered Reports as “a publishing format…that emphasizes the importance of the research question and the quality of methodology by conducting peer review prior to data collection”. In practice this means changing the way research is conducted and published.
Background and benefits
A Registered Report is a form of empirical article offered by the journal. The methods and proposed analyses are submitted to the journal for review prior to research being conducted. High-quality protocols are then provisionally accepted for publication and are pre-registered before data collection commences.
This format is designed to improve research design while minimizing publication bias other issues in hypothesis-driven research. It allows for the flexibility to conduct exploratory (unregistered) analyses and report unplanned findings.
Registered Reports aim to reduce questionable research practices such as P-hacking, Cherry Picking, HARKing.
Publishing tips, direct to your inbox
Expert tips and guidance on getting published and maximizing the impact of your research. Register now for monthly insights direct to your inbox.
Help with your Registered Report submission
Interested in submitting a registered report but don’t know where to start? We’ve got you covered. Get detailed information on submitting a Registered Report.
Explore journals offering Registered Reports
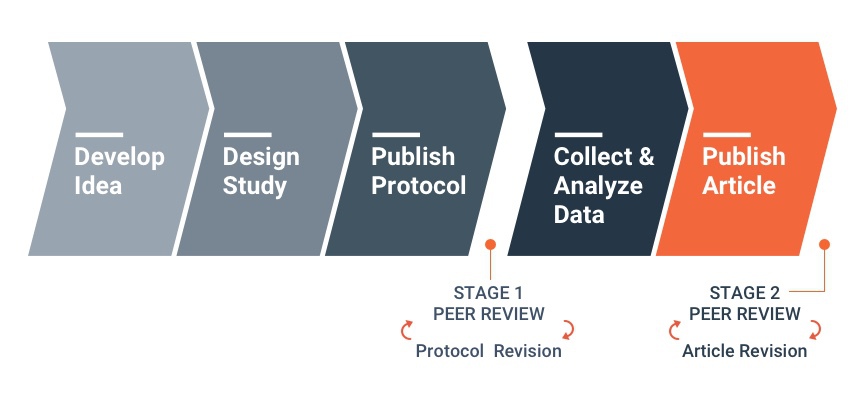
How Registered Reports work

The review process for Registered Reports is divided into two stages. At Stage 1, reviewers assess study proposals before data are collected. At Stage 2, reviewers consider the full study, including results and interpretation.
Stage 1 manuscripts will include only an ‘introduction’, ‘methods’ (including proposed analyses), and ‘pilot data’ (where applicable). In considering papers at stage 1, reviewers will be asked to assess:
The importance of the research question(s).
The logic, rationale, and plausibility of the proposed hypotheses.
The soundness and feasibility of the methodology and analysis pipeline (including statistical power analysis where appropriate).
Whether the clarity and degree of methodological detail is sufficient to exactly replicate the proposed experimental procedures and analysis pipeline.
Whether the authors have pre-specified sufficient outcome-neutral tests for ensuring that the results obtained are able to test the stated hypotheses, including positive controls and quality checks.
Following Stage 1 peer review, manuscripts will be accepted, offered the opportunity to revise, or rejected outright. Manuscripts that pass peer review will be issued an in principle acceptance (IPA), indicating that the article will be published pending successful completion of the study according to the pre-registered methods and analytic procedures, as well as a defensible and evidence-based interpretation of the results.
Following completion of the study, authors will complete the manuscript, including results and discussion sections. These stage 2 manuscripts will more closely resemble a regular article format. The manuscript will then be returned to the reviewers, who will be asked to appraise whether:
The data are able to test the authors’ proposed hypotheses by satisfying the approved outcome-neutral conditions (such as quality checks, positive controls).
The ‘introduction’, rationale and stated hypotheses are the same as the approved stage 1 submission (required).
The authors adhered precisely to the registered experimental procedures.
Any unregistered post hoc analyses added by the authors are justified, methodologically sound, and informative.
The authors’ conclusions are justified given the data.
Reviewers at stage 2 may suggest that authors report additional post hoc tests on their data; however, authors are not obliged to do so unless such tests are necessary to satisfy one or more of the stage 2 review criteria.

Please note that editorial decisions will not be based on the perceived importance, novelty, or conclusiveness of the results.
Registered Reports on F1000Research
F1000Research welcomes Registered Reports, but follows a slightly different model where both the Study Protocol and the consequent Research Article are published and peer-reviewed. Most journals typically only publish the Research Article, so the F1000Research approach to Registered Reports rewards authors with an additional citable publication, and goes further in supporting transparent, reproducible research.