Guidance on submitting a Registered Report
Have your study peer reviewed and get an in-principle acceptance before you start collecting your data
How to submit a Registered Report
Want to submit a Registered Report but don’t know where to start? We will guide you through the submission and review process and provide you with the tools you need.
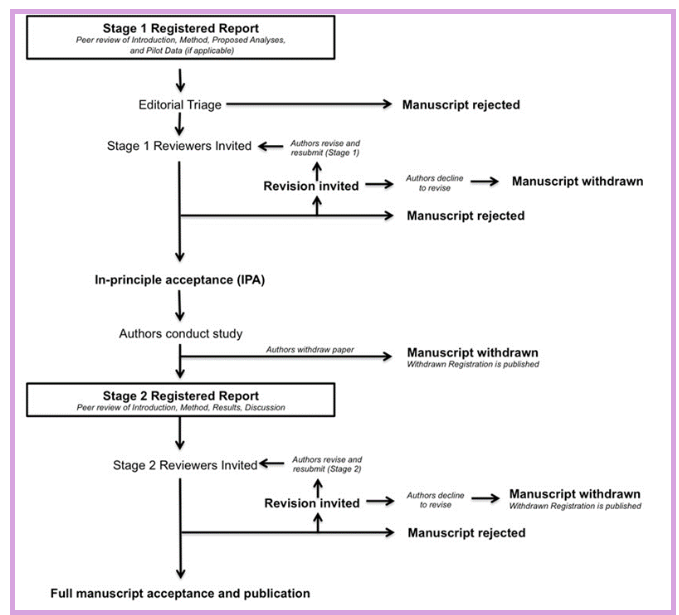
The review process
In this section we explain the review process for Stage 1 and for Stage 2 in full. We also have some downloadable cover letters that you can make use of.
Publishing tips, direct to your inbox
Expert tips and guidance on getting published and maximizing the impact of your research. Register now for monthly insights direct to your inbox.

Stage 1 Registered Report
Where journals receive high levels of submissions annually, not all manuscripts will automatically proceed to peer review. Editors may only select reviewers for the most promising manuscripts. Therefore we have provided you with what editors will look for.
Stage 1 submissions should include the sections outlined below and a brief cover letter. Authors are welcome to submit pre-submission enquiries for advice on the likely suitability of a study as a Registered Report. However, please note that the editors will not agree to send manuscripts for in-depth review until a complete Stage 1 submission has been received.
Stage 1 submissions that are judged by the editors to be of sufficient quality and scientific importance will be sent for in-depth peer review. In considering papers at the registration stage, reviewers will be asked to assess the following:
The importance of the research question(s).
The logic, rationale, and plausibility of the proposed hypotheses.
The soundness and feasibility of the methodology and analysis pipeline (including statistical power analysis where appropriate).
Whether the clarity and degree of methodological detail is sufficient to exactly replicate the proposed experimental procedures and analysis pipeline.
Whether the authors have pre-specified sufficient outcome-neutral tests for ensuring that the results obtained are able to test the stated hypotheses, including positive controls and quality checks.

Following Stage 1 peer review, manuscripts may be rejected outright, offered the opportunity to revise, or get accepted. In principle acceptance (IPA) indicates that your article will be published as long as you successfully complete your study according to the pre-registered methods and submit an evidence-based interpretation of the results. Stage 1 protocols are not published following IPA. Instead, they are held in reserve by the journal and integrated into a single completed article following approval of the final Stage 2 manuscript.
Please note: Authors are encouraged to follow the stated experimental procedures, as any deviation, regardless of how minor it may seem, could lead to rejection of the manuscript at Stage 2.
In cases where the pre-registered protocol is altered after IPA due to unforeseen circumstances (e.g. change of equipment or unanticipated technical error), the authors must consult the editors immediately for advice, and prior to the completion of data collection. Minor changes to the protocol may be permitted according to editorial discretion. In such cases, IPA would be preserved, and the deviation reported in the Stage 2 submission. If authors wish to alter the experimental procedures more substantially following IPA but still wish to publish their article as a Registered Report, the manuscript must be withdrawn and resubmitted as a new Stage 1 submission.
Note that all registered analyses must be undertaken and reported in the Stage 2 manuscript, but additional unregistered analyses may also be included in a final manuscript (see Results & Discussion).
Stage 1 cover letter
A brief scientific case for consideration. Authors are encouraged to refer to the likely replication value of the research. High-value replication studies are welcome in addition to novel studies.
An anticipated timeline for completing the study if the initial submission is accepted.
How to structure your Stage 1 Manuscript
Introduction
The introduction should be a review of the relevant literature that motivates the research question and a full description of the experimental aims and hypotheses. Please note that following IPA, the Introduction section cannot be altered except for correction of typographical errors (see below).
Methods
The method section is a full description of proposed sample characteristics, including criteria for data inclusion and exclusion (e.g. outlier extraction). Procedures for objectively defining exclusion criteria due to technical errors or for any other reasons must be specified, including details of how and under what conditions data would be replaced.
A description of experimental procedures in sufficient detail to allow another researcher to repeat the methodology exactly, without requiring further information. These procedures must be adhered to exactly in the subsequent experiments or any Stage 2 manuscript can be rejected.
Proposed analysis pipeline, including all pre-processing steps, and a precise description of all planned analyses, including appropriate correction for multiple comparisons. Any covariates or regressors must be stated. Where analysis decisions are contingent on the outcome of prior analyses, these contingencies must be specified and adhered to. Only pre-planned analyses can be reported in the main Results section of Stage 2 submissions. However, unplanned exploratory analyses will be admissible in a separate section of the Results (see below).

Studies involving Neyman-Pearson inference must include a statistical power analysis. Estimated effect sizes should be justified with reference to the existing literature. Since publication bias overinflates published estimates of effect size, power analysis must be based on the lowest available or meaningful estimate of the effect size. For frequentist analysis plans, the a priori power must be 0.9 or higher for all proposed hypothesis tests. In the case of highly uncertain effect sizes, a variable sample size and interim data analysis is permissible but with inspection points stated in advance, appropriate Type I error correction for ‘peeking’ employed, and a final stopping rule for data collection outlined.
Methods involving Bayesian hypothesis testing are encouraged. For studies involving analyses with Bayes factors, the predictions of the theory must be specified so that a Bayes factor can be calculated. Authors should indicate what distribution will be used to represent the predictions of the theory and how its parameters will be specified. For example, will you use a uniform up to some specified maximum, or a normal/half-normal to represent a likely effect size, or a JZS/Cauchy with a specified scaling constant?
For inference by Bayes factors, authors must be able to guarantee data collection until the Bayes factor is at least 6 times in favour of the experimental hypothesis over the null hypothesis (or vice versa). Authors with resource limitations are permitted to specify a maximum feasible sample size at which data collection must cease regardless of the Bayes factor; however, to be eligible for advance acceptance this number must be sufficiently large that inconclusive results at this sample size would be an important message for the field. For further advice on Bayes factors or Bayesian sampling methods, prospective authors are encouraged to read this key article by Schönbrodt and Wagenmakers.
Full descriptions must be provided of any outcome-neutral criteria that must be met for successful testing of the stated hypotheses. Such quality checks might include the absence of floor or ceiling effects in data distributions, positive controls, or other quality checks that are orthogonal to the experimental hypotheses.
Timeline for completion of the study and proposed resubmission date if Stage 1 review is successful. Extensions to this deadline can be negotiated with the editors. Manuscripts will be generally considered only for studies that are able to commence immediately; however, authors with alternative plans are encouraged to contact the journal office for advice.
Any description of prospective methods or analysis plans should be written in future tense.
Pilot Data (optional):
Can be included to establish proof of concept or feasibility of proposed methods. Any pilot experiments will be published with the final version of the manuscript and will be clearly distinguished from data obtained for the pre-registered experiment(s).
Secondary Registrations (optional):
The journal welcomes submissions proposing secondary analyses of existing data sets, provided authors can supply sufficient evidence (e.g. letter from independent gatekeeper) to confirm that they have had no prior access to the data in question.
Stage 2 Registered Report
Once the study is complete, you can prepare and resubmit your manuscript for full review. The final manuscript should include the entirety of the Stage 1 submission, the raw data and laboratory logs, and the results and discussion. Apart from minor stylistic revisions, the Stage 1 submission cannot be altered.
At Stage 2, any description of the rationale or proposed methodology that was written in future tense within the Stage 1 manuscript should be changed to past tense. Any textual changes to the ‘introduction’ or ‘methods’ (e.g. correction of typographic errors) must be clearly marked in the Stage 2 submission. Any relevant literature that appeared following the date of IPA should be covered in the ‘discussion’.
Reviewers are informed that editorial decisions will not be based on the perceived importance, novelty or conclusiveness of the results. While reviewers are free to enter such comments on the record, they will not influence editorial decisions. Reviewers at Stage 2 may suggest that authors report additional post hoc tests on their data; however, authors are not obliged to do so unless such tests are necessary to satisfy one or more of the Stage 2 review criteria.
Stage 2 Cover Letter
Please note the policy of publishing negative results under the Registered Reports scheme does not imply that the journal publishes negative results for other manuscript types.
The resubmission will most likely be considered by the same reviewers as in Stage 1 but could also be assessed by new reviewers. In considering papers at Stage 2, reviewers will be asked to decide:
If the data are able to test the authors’ proposed hypotheses by satisfying the approved outcome-neutral conditions (such as quality checks, positive controls).
If the Introduction, rationale and stated hypotheses are the same as the approved Stage 1 submission (required).
If the authors adhered precisely to the registered experimental procedures.
If any unregistered post hoc analyses added by the authors are justified, methodologically sound, and informative.
If the authors’ conclusions are justified given the data.
Raw data and laboratory log
Submission of raw data and laboratory log: Share Your Data

Results & Discussion

The outcome of all registered analyses must be reported in the manuscript, except in rare instances where a registered and approved analysis is subsequently shown to be logically flawed or unfounded. In such cases, the authors, reviewers, and editors must agree that a collective error of judgment was made, and that the analysis is inappropriate. In such cases the analysis would still be mentioned in the ‘methods’ but omitted with justification from the ‘results’.
It is reasonable that you may wish to include additional analyses that were not included in the registered submission. For instance, a new analytic approach might become available between IPA and Stage 2 review, or a particularly interesting and unexpected finding may emerge. Such analyses are admissible but must be clearly justified in the text, appropriately caveated, and reported in a separate section of the Results titled “Exploratory analyses”. You should be careful not to base their conclusions entirely on the outcome of statistically significant post hoc analyses. Authors reporting null hypothesis significance tests are required to report exact p values and effect sizes for all inferential analyses.
Manuscript withdrawal and withdrawn registrations
It is possible that authors with IPA may wish to withdraw their manuscript following or during data collection. Possible reasons could include major technical error, an inability to complete the study due to other unforeseen circumstances, or the desire to submit the results to a different journal. In all such cases, manuscripts can of course be withdrawn at the authors’ discretion. However, the journal will publicly record each case in a section called Withdrawn Registrations.
This section will include the authors, proposed title, the abstract from the approved Stage 1 submission, and brief reason(s) for the failure to complete the study. Partial withdrawals are not possible; i.e. authors cannot publish part of a registered study by selectively withdrawing one of the planned experiments. Such cases must lead to withdrawal of the entire paper. Studies that are not completed by the agreed Stage 2 submission deadline (6 months after receipt of the IPA – this can be extended in negotiation with the editors) will be considered withdrawn and will be subject to a Withdrawn Registration.

Incremental Registrations

You can choose to add experiments to approved submissions. In such cases the approved Stage 2 manuscript will be accepted for publication, and authors can propose additional experiments for Stage 1 consideration. Where these experiments extend the approved submission (as opposed to being part of new submissions), the editors will seek to fast-track the review process. This option may be particularly appropriate where an initial experiment reveals a major serendipitous finding that warrants follow-up within the same paper.
In cases where an incremented submission is rejected (at either Stage 1 or 2), authors will retain the option of publishing the most recently approved version of the manuscript. For further advice on specific scenarios for incremental registration, authors are invited to contact the editors.